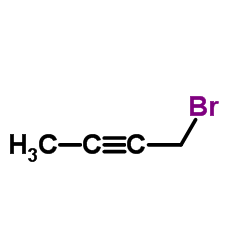
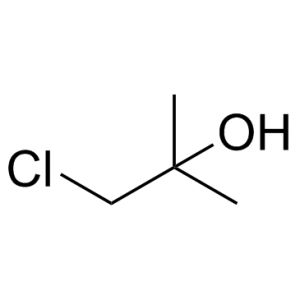
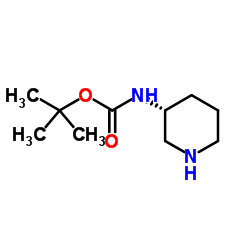
8-[(3R)-3-aminopiperidin-1-yl]-7-but-2-ynyl-3-methyl-1-[(4-methylquinazolin-2-yl)methyl]purine-2,6-dione
| Product Name | Linagliptin CAS NO668270-12-0 |
| Synonyms | (R)-8-(3-aminopiperidin-1-yl)-7-(but-2-yn-1-yl)-3-Methyl-1-((4-Methylquinazolin-2-yl)Methyl)-1H-purine-2,6(3H,7H)-dione; |
| CAS No | 668270-12-0 |
| Molecular Formula | C25H28N8O2 |
| Molecular Weight | 472.542 |
1. Linagliptin(BI-1356) is an inhibitor of DPP-4, an enzyme that degrades the incretin hormones glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP).
2. 8-[(3R)-3-Amino-1-piperidinyl]-7-(2-butyn-1-yl)-3-methyl-1-[(4-methyl-2-quinazolinyl)methyl]-3,7-dihydro-1H-purine-2,6-dione is an novel potent and selective dipeptidyl peptidase-4 (DPP-4) inhibitor with potential use in the treatment of type 2 diabetes.
Our factory manufacture’s Linagliptin intermediate CAS 3355-28-0, mature technology, stable output, quality assurance.
Inventory Status : In Stock.
If you are interested in our products or have any questions, please feel free to contact us!
Products under patent are offered for R & D purpose only.
Use: It is a potent, selective DPP-4 inhibitor with a IC50 value of 1 nM.
In vitro study: Ligagliptin inhibited DPP-4 activity in vitro in several independent experiments with IC50 values of 0.4,0.5,0.9 and 1.1nM (mean IC50, approximately 1nM). The IC50 of FAP inhibition by liagliptin was 89 nM (about 90-fold selectivity relative to DPP-4).
In vivo research: In male Wistar rats, Beagle dogs and rhesus monkeys, xanthine linagliptin was shown to be an efficient, durable and potent DPP-4 inhibitor with> 7 h> 70% / kg after 1 mg of oral administration. Oral administration of ritagliptin to db / db mice alone, 45 min before the oral glucose tolerance test, dose-dependently reduced the plasma glucose shift from 0.1mg / kg (15% inhibition) to 1mg / kg (66% inhibition) [1]. Ligagliptin (3 and 10mg / kg) dose-dependently inhibited the DPP-4 enzyme in the plasma within 30 min of drug administration. Ligagliptin (1 mg / kg, po) significantly reduced glucose offset by about 50% [2]. Oral administration of the DPP-4 inhibitor ligagliptin (3mg / kg, oral administration) strongly reduced DPP-4 activity, stabilized active GLP-1 in chronic wounds, and improved healing in ob / ob mice. At day 10 post-injury, liagliptin-treated ob / ob mice showed mostly epithelialized wounds, characterized by the absence of neutrophils.
Document: A new chemical class of potent DPP-4 inhibitors structurally derived from the xanthine scaffold for the treatment of type 2 diabetes has been discovered and evaluated. Systematic structural variations have led to 1 (BI 1356), a highly potent, selective, long-acting, and orally active DPP-4 inhibitor that shows considerable blood glucose lowering in different animal species. 1 is currently undergoing clinical phase IIb trials and holds the potential for once-daily treatment of type 2 diabetics.




![8-[(3R)-3-aminopiperidin-1-yl]-7-but-2-ynyl-3-methyl-1-[(4-methylquinazolin-2-yl)methyl]purine-2,6-dione Featured Image](http://cdn.globalso.com/cheerour/dadas1.png)